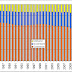
Today’s Managing Health Care Costs Indicator is 8.5%
Click to enlarge image
The AARP reported last week that brand name prescription drug prices rose 8.5% last year. That’s substantially more than the total increase in the cost of pharmaceuticals - which was 3.2% for 2008 (last publication of National Health Expenditures or 5.7% (CVS-Caremark- figure above).
What’s the difference?
The overwhelming majority of prescriptions (about 70%) are for generic medications. However, the overwhelming majority of costs (about 80%) are for that minority of medications that are available in brand name formulations only. So – generic substitution can lead to giant savings, but these can also be wiped out quickly when brand name prices go up.
The big pharmas aren’t in an enviable position. Many ‘blockbuster’ drugs have gone generic over the past few years, and Pfizer is preparing itself for Lipitor (atorvastatin) going generic at the end of next year. The AARP study shows that drugs about to go generic have had large cost increases in recent years. Why is this?
It’s not to fund the research (long since paid for), but it could help underwrite new research. It’s not to pay for marketing – the pharmas cease large-scale promotion of a drug in the months before it goes generic. Brand name price increases are certainly not to address additional production costs – generic manufacturers will be able to produce these for pennies a pill in just a few months.
The pharmas cannot suggest an increase in ‘value’ provided by a brand name as it nears patent expiry which could justify an increase often over 10%.
Once again –what causes the prices of drugs about to go generic to increase?
There are a few different underlying reasons. The first is revenue maximization. The ability to get a high margin is about to evaporate – make hay while the sun shines! A darker potential reason is that the pharmas are seeking to make drugs about to go off patent look as unattractive as possible. When Lipitor is about to lose its patent protection, even while a similar drug simvastatin is available for pennies a pill , Lipitor goes up in price by 24%. Sure, that will move some patients to simvastatin. But that will diminish profits for the ‘first mover’ generic competitor, as some patients will switch to another statin medication that retains patent protection. Flomax went up 92% over 5 years; most of that price increase was in the two years before patent expiration.
Many pharmacy benefit managers would move drugs about to lose their patent protection to a “preferred” tier to gain savings after the drug goes generic. However, the manufacturers would like patients to move away from drugs about to lose their patent protection – preferably to a medication that will retain patent protection for a number of additional years.
There aren’t perfect answers here. Most other developed countries have rigorous price controls for pharmaceutical agents, but this can stifle innovation. Clearly, “me too” drugs can offer an opportunity to create some price competition, which helps improve value. The pharmas used to introduce controlled release formulations for a drug about to lose patent protection; we have seen fewer of these end-runs around generic substitution recently. New regulations to make it more difficult for the brand name pharmas to pay a generic manufacturer for a delay can drive some additional volume to the lower-cost generic earlier.
Click to enlarge image